| |  | | | | | | Did someone forward this newsletter to you? Sign up here to get it in your inbox. In today’s issue: - The Trump administration closed a deal with Regeneron, completing drug pricing deals with 17 major drugmakers
- Drugmakers overall are ramping up lobbying amid a bipartisan interest in codifying the principles of the pricing agreements into law
- Regulators announce new pathway to speed Medicare coverage for certain medical devices
… and more. This is the Health Brief newsletter. And thanks for joining us on this gorgeous Thursday. We’ve covered lobbying spending in early 2026 among top hospital industry players, now let’s check in with drugmakers. Do you have any health policy tips or scoops? Are you headed to any White House correspondents’ dinner festivities in the coming days? Don’t hesitate to reach out. I’m at megan.wilson@washpost.com. Or, if you’d prefer to message me securely, I’m also on Signal at megan. 434. This newsletter is published by WP Intelligence, The Washington Post’s subscription service for professionals that provides business, policy and thought leaders with actionable insights. WP Intelligence operates independently from The Washington Post newsroom. Learn more about WP Intelligence. |  | | Regeneron CEO Leonard Schleifer speaks at the White House on Thursday while announcing a drug-pricing deal with the Trump administration. (Brendan Smialowski/AFP, Getty Images) | | | |  | The Lead Brief | The Trump administration has struck a deal with Regeneron Pharmaceuticals to align some of its drug prices with those paid in other wealthy countries — marking the final piece of the White House’s push to lock in “most-favored-nation” pricing agreements with some of the world’s largest drugmakers. With Regeneron on board, President Donald Trump has now secured agreements with all 17 major drugmakers he targeted in letters last year — including Pfizer, BMS, Eli Lilly and GSK — ordering companies to lower prices on their products. Read more: Dan Diamond in The Washington Post newsroom has all the details about the deal, which provides Regeneron with tariff relief and other incentives. The company said it would be offering its newly approved gene therapy, which targets genetic hearing loss, free in the United States. The individual deals, which remain a secret, have become a flash point for Democrats looking to push back on a key part of the administration’s affordability narrative ahead of the midterm elections. Lawmakers on Capitol Hill skeptical of the scope of the purported savings in the deals have been seeking more information about them. → The announcement Thursday comes as some of the drugmakers who’ve cut deals with the administration — and their smaller industry counterparts — are fiercely lobbying to keep Congress from heeding the Trump administration’s call to codify the principles underlying the deals. Even though many Republicans on Capitol Hill are skeptical of pushing most-favored-nation pricing policies, viewing the proposal as government-imposed price controls, the pharmaceutical industry will need to remain vigilant. The idea of tying U.S. drug prices to lower prices abroad has support from many Democrats. Health Secretary Robert F. Kennedy Jr. offered to work with Democratic senators on the legislative effort to codify the principles of the administration’s drug-pricing deals. → On Wednesday, a proposal from Sen. Bernie Sanders (I-Vermont) to add an MFN pricing amendment to the Senate Republicans’ budget resolution failed to advance — but garnered “yes” votes from Sens. Susan Collins (R-Maine), Josh Hawley (R-Missouri) and Dan Sullivan (R-Alaska). Collins and Sullivan are currently in competitive races to keep their seats. | | | |  | Lobbying Ledger | Of the companies that cut deals with the administration, 11 increased their advocacy spending in the beginning of this year compared to the same time last year. Here are the largest lobbying spending increases among companies that cut deals with the Trump administration: | 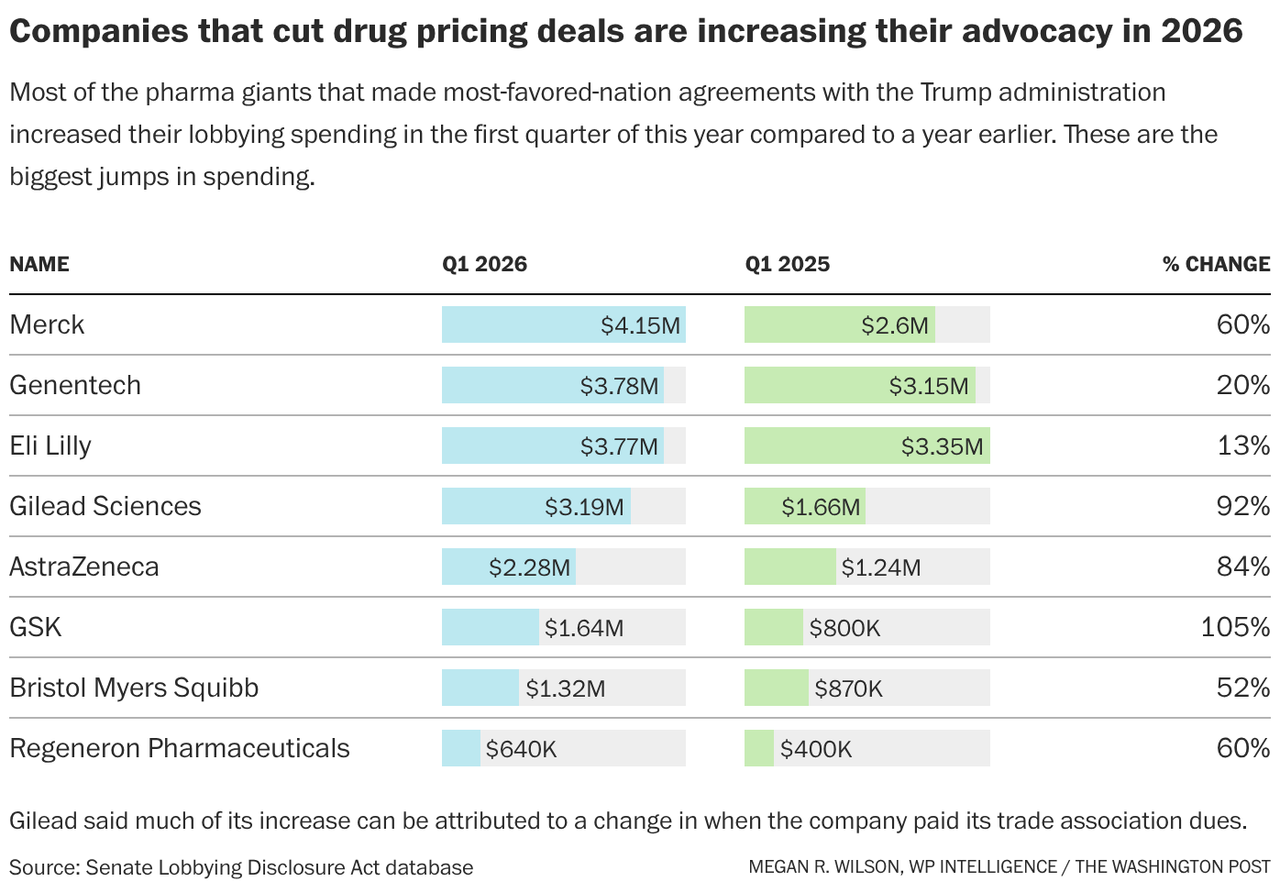 | | In the first quarter of 2026, Regeneron spent $640,000 on lobbying, the highest quarterly amount since it started engaging in advocacy nearly 14 years ago. Regeneron — which listed working on issues including drug pricing, intellectual property legislation and tariffs on disclosure forms — didn’t respond to a request for comment about its lobbying spending. It has four outside lobbying firms on retainer, including Checkmate Government Relations. “We were not pushed to be here. We are happy to be here because it marks an important step to lower drug prices,” Regeneron CEO Leonard Schleifer said Thursday during an Oval Office announcement. “We’ve been arguing for more than a decade that other wealthy nations have been getting a free ride on American innovation.” AstraZeneca, which became the second company to announce a drug-pricingdeal with the Trump administration in October, also broke its own lobbying spending records this year, according to a review of federal records. The drugmaker spent about $2.3 million on lobbying during the first quarter of 2026, the most it has ever spent on lobbying in any quarter. AstraZeneca said that its increase in lobbying spending and interactions with Washington reflect the company’s growing presence in the United States. Its disclosures mention advocacy work on drug pricing, tariffs, changes to the 340B discount drug program and the reauthorization of a pediatric priority review voucher program that incentivizes the development of rare-disease drugs. It has four outside lobbying firms on retainer. Eli Lilly’s lobbying spending also reached a record high in the first three months of this year. The company shelled out nearly $3.8 million on advocacy, a 13 percent increase over the same period in 2025 but more than double its lobbying spending in the first quarter of 2024. Its disclosure forms mention working on a broad swath of topics, including drug pricing and reimbursement issues, the use of artificial intelligence in health care, and prescription drug approvals. It has 15 outside lobbying firms on retainer. The company did not respond to a request for comment about its lobbying spending. In November, Eli Lilly announced a drug-pricing deal with the Trump administration that included an agreement to lower prices on its popular GLP-1 weight-loss drugs in exchange for tariff relief, expedited regulatory review of a new weight-loss medication and broader access to the Medicare program. Gilead Sciences spent $3.19 million in the first quarter of 2026, nearly double its lobbying expenditures from the same time last year. A spokesperson said the increase can largely be attributed to a change in when it had paid its membership dues to industry groups such as Pharmaceutical Research and Manufacturers of America and the Biotechnology Innovation Organization. | | | |  | Industry Rx | Overall, there were 38 branded drug companies that each spent at least $460,000 on lobbying in the first quarter of 2026. Most increased their spending compared to the same period in 2025, with a median jump of nearly 40 percent over last year, according to my analysis of federal records. Midsize biopharmaceutical companies have been ramping up their engagement with Washington, as the industry has expressed frustration with the drug-approval process, and amid fears they could get swept into the Trump administration’s drug-pricing push. Many argue that they don’t have the flexibility to offer price concessions as larger manufacturers do. The Biotechnology Innovation Organization, whose membership includes hundreds of small and midsize pharma companies, called the effort to codify the MFN policies into law “the greatest threat facing our industry.” BIO’s chief executive, John F. Crowley, recently told members that the group is “actively advocating against these proposals.” He argues that would disincentivize investment, shrink the number of drugs companies are able to develop and “damage the free market conditions that make the U.S. the global engine of biomedical innovation.” The group spent more than $1.6 million on lobbying in the first quarter of 2026, an 82 percent increase over the same period in 2025. The figure, however, is on par with its lobbying spending in the first quarter of 2024. The organization uses a more expansive reporting method that includes spending on other advocacy, including state lobbying, that can translate to higher spending in election years. UCB, which increased its first-quarter lobbying 160 percent compared to the same time in 2025, wouldn’t comment on what spurred the jump. However, the only issue listed on its internal lobbying disclosure for the most recent quarter is simply: “Tariffs.” The company touted its recent announcement that it plans to build a new manufacturing facility in Georgia. Increasing domestic investment has been a major priority for Trump, who has offered companies breaks from tariffs if they expand their operations in the U.S. UCB spent $520,000 on lobbying during the first three months of this year, its second-highest quarterly spending figure after second-quarter 2025. It has three lobbying firms on retainer. “This is a rapidly changing environment,” said company spokesperson Erica Puntel. “We remain committed to working constructively with policymakers on solutions that support patient access to innovative medicines.” Biogen spent nearly $1.5 million in the first quarter of 2026, a 43 percent increase over the same period in 2025 and a 38 increase over first-quarter 2024. The company told me that much of the disclosed lobbying increases are due to a change in the timing of its trade association membership dues. Although a Biogen spokesperson added that there hasn’t been a “material change” to its overall approach to policy engagement in the last year, the company acknowledged that its overall advocacy has ramped up in recent years. “As Biogen has grown over the past few years and expanded research and development into new disease areas, this growth equates to growth at the public policy level,” the spokesperson said in an email. | | | |  | Executive Health Brief | Patients may get speedier Medicare coverage of some new medical devices under a Trump administration initiative announced Thursday, Rebecca Adams, the lead health care analyst at WP Intelligence, reports. The Centers for Medicare and Medicaid Services and the Food and Drug Administration plan to work together to tackle a key bottleneck for certain devices deemed breakthrough technologies — defined as offering more effective treatment for life-threatening or irreversibly debilitating conditions. The issue: Even after the FDA signs off on a new medical device, obtaining Medicare coverage can take a year or more, in part because CMS officials often request additional data that wasn’t required for clearance. That lag forces manufacturers into costly extra studies and can slow coverage across the broader market, since private insurers often follow Medicare’s lead. The medical device industry has been asking regulators to help streamline the process, which is part of what prompted the changes, Grace Graham, a top FDA official, told reporters Thursday. WHAT’S NEW - Under the new pathway, called the Regulatory Alignment for Predictable and Immediate Device, or RAPID, the two agencies say they will begin coordinating earlier in the process and will inform device manufacturers up front what evidence is needed for both FDA clearance and Medicare coverage.
- The more streamlined approach could provide national Medicare coverage as soon as two months after the FDA authorizes a device.
- High-risk breakthrough medical devices and some moderate-risk devices could qualify for the coordinated reviews if companies enroll Medicare patients in trials that show benefits identified by both agencies. In practice, that means the category of devices that might be available sooner includes artificial heart valves, devices that treat heart rhythms or implants that stimulate nerves to treat disease.
Industry reaction: “While we appreciate this first step toward improving coverage, it is critical that the implementation of such a proposal is effective,” said Scott Whitaker, the president and CEO of the medical technology association AdvaMed. “Without meaningful timelines, accountability and effective management of this program, patients are unlikely to see the full benefits of new technologies.” What to watch: The House Ways and Means Committee in September approved, by a resounding 37-3 vote, a bill to create a four-year temporary Medicare coverage path for breakthrough devices upon FDA approval. The manufacturer and CMS would then work together to establish permanent coverage. → On Wednesday, a bipartisan group of 82 lawmakers sent the Department of Health and Human Services a letter calling for faster coverage. By the numbers: The Trump administration anticipates about 40 breakthrough medical devices to be covered in the beginning, and then about 20 more devices every year. That’s far more than a Biden administration-era program that capped the number at five annually. | | | |  | From our notebook | | | | | | | | | |