 | | | | |  | By Megan R. Wilson | Did someone forward this newsletter to you? Sign up here to get it in your inbox. In today’s issue: - The next sector in health care that could find itself under the microscope on Capitol Hill
- Senate Democrats seek more information about whether the agreements drug companies cut with the Trump administration will result in greater savings to Medicaid than the discounts already in the program
- New polling shows who Americans trust for public health information over both the Centers for Disease Control and Prevention and the administration’s top health officials
- Another front opens in the battle between hospitals and drugmakers over the 340B drug discount program
… And more. Good afternoon, and thanks for checking in with the Health Brief newsletter. Please send any tips or leads to megan.wilson@washpost.com. If you prefer to message me securely, I’m also on Signal at megan. 434. This newsletter is published by WP Intelligence, The Washington Post’s subscription service for professionals that provides business, policy and thought leaders with actionable insights. WP Intelligence operates independently from The Washington Post newsroom. Learn more about WP Intelligence. | | | |  | The Lead Brief | The supply chain that ultimately connects patients with their medicines is winding and complex — and growing more consolidated. In recent years, lawmakers have targeted the tactics used by drug companies and pharmacy benefit managers, or PBMs, to expand their businesses and bolster their revenue, but there’s another sector that’s been flying under the radar: drug distributors. My WP Intelligence colleague Rebecca Adams takes a look at this part of the health care world in her latest in-depth report. The three top wholesale drug distributors — McKesson, Cencora and Cardinal Health — aren’t household names, but they control at least 96 percent of the nation’s drug delivery system. They’re also Fortune 15 companies, among the largest companies in the U.S. by revenue, putting them in the same league as JPMorgan Chase and Microsoft. Drug distributors serve a range of customers, including hospitals, clinics, long-term care facilities and drugstores. These companies move more than 10 million medicines from 1,400 manufacturers to providers every day, according to the Healthcare Distribution Alliance, the association representing the industry. The wholesalers are increasingly diversifying far beyond their traditional role of warehousing and delivering drugs, and lawmakers are taking notice. Rep. Kat Cammack (R-Florida) pointed out at a recent congressional hearing that distributors have spent $16 billion acquiring or partnering with physician management service companies that invest in doctors’ practices. They have also invested in specialty pharmacies, group purchasing organizations and other businesses. Why it matters: Physician practices are less able to shop around for lower-priced drugs when a wholesaler is an investor in those same practices. (An area where this is particularly acute, according to Rebecca’s report, is oncology centers.) Overall, the complex ties make it even less likely that doctors’ offices will skip the wholesaler middleman to buy drugs directly from manufacturers. The cemented connections also make it harder for other wholesalers to compete for business. The relationships also raise questions about whether investments from a wholesaler in physician practices influence doctors’ treatment decisions. → The industry is pushing back on these concerns. The Healthcare Distribution Alliance told Rebecca that pharmaceutical distributors “do not influence clinical decisions, do not set the prices that patients pay, and do not determine what medicines are available through a patient’s formulary.” “Those decisions are made by other health care stakeholders,” the association said. “Our sector is working to build the most resilient and efficient supply chain to the benefit of the patients.” House Energy and Commerce Committee Chairman Brett Guthrie (R-Kentucky) questioned whether integration is good for the health care system, pointing to how the three largest companies represent most of the industry’s market power. Chip Davis, the Healthcare Distribution Alliance’s president and CEO, told lawmakers that the “Big Three” distributors do not fully own medical practices. Davis said that his association’s members are building out diversified business practices, in part because they are hearing from customers about their inability to access affordable medication through other sources. What to watch: While it’s unlikely that lawmakers will move legislation that tackles industry consolidation or the rising cost of health care in the near term, the discussion has gathered bipartisan support. It took years for Congress to pass PBM reforms that were recently enacted in a government funding bill, and other overhauls could take time. The criticism from Republicans at the House Energy and Commerce Committee drug affordability hearing last month, combined with Senate Democrats calling out distributors in a letter outlining policy proposals, could mean that legislation could be possible down the road. — With assistance from Rebecca Adams | | | |  | Document Drop | The American Economic Liberties Project, a nonprofit that advocates for bolstered antitrust enforcement, has a new report that calls for more onshoring of the drug supply chain, Rebecca reports. → The group says that wholesalers use their market power to reduce the profits of domestic generic drug companies. The report highlights the potential risks for Americans when generic drug manufacturing occurs in low-cost countries overseas. → For its part, the Healthcare Distribution Alliance has its own set of policy proposals to shore up the generic drug market. | | Sen. Ron Wyden (Oregon), the top Democrat on the Senate Finance Committee, is asking drugmakers to detail the pricing deals they've made with the Trump administration. (Maansi Srivastava/for the Washington Post) | | | |  | Health on the Hill | Sen. Ron Wyden (Oregon), the Senate Finance Committee’s top Democrat, wants to know whether states are getting better deals on medications through the Trump administration’s drug pricing deals or via the deep discounts already provided through Medicaid. Companies have agreed to provide “most-favored nation” pricing on certain products in Medicaid as part of those deals, which the administration is implementing through a voluntary pilot program known as the GENErating cost Reductions fOr U.S. Medicaid, or GENEROUS, model. But, in letters sent to the 16 companies that cut deals including promises to lower the costs of their drugs in line with wealthy countries, Wyden and six other Democrats are asking whether those net prices are the same — or higher — than what Medicaid already pays. → The letters also ask companies to spell out in more detail which of their products will be covered under the deals in the Medicaid program, which is jointly administered by the federal government and states. “Critical details about the GENEROUS model also remain unclear, including whether manufacturers can seek exemptions for the drugs where this model could have the greatest impact,” read the letters, also signed by Democratic Sens. Amy Klobuchar (Minnesota), John Hickenlooper (Colorado), Elizabeth Warren (Massachusetts), Dick Durbin (Illinois), Jeff Merkley (Oregon) and Chris Murphy (Connecticut). Why it matters: The letters are part of a broader inquiry by Wyden and the panel’s Democrats, who are trying to learn about the administration’s drug pricing agreements — which are confidential — and whether they will actually result in lower medicine costs. (Wyden has also partnered with House Democrats on other letters.) → In addition to the correspondence, Democrats on the Senate Finance Committee have been meeting with the companies that have cut deals with the administration in an effort to learn more about the agreements and how they came together. What to watch: Although Democrats are in the minority, they are laying the groundwork to advance policy priorities — including building on the Medicare negotiation program set up by the Inflation Reduction Act — should they regain control of one or both chambers in November’s midterm elections. Last month, Wyden led a “Dear Colleague” letter asking for input from lawmakers on plans to advance additional measures aiming to tackle high medicine costs. He said the committee will “dedicate substantial time and effort this year” into coming up with proposals to lower health care costs — including proposals to expand Medicare’s negotiation program and creating incentives for drugmakers to research and develop new medicines. | | | |  | Numbers Game | A new poll from the University of Pennsylvania’s Annenberg Public Policy Center reveals that Americans are more likely to accept vaccine recommendations from the American Academy of Pediatrics or the American Medical Association than from the Centers for Disease Control and Prevention. → While about 60 percent of the people surveyed said they trusted the CDC’s information about the safety and efficacy of vaccines, they also reported leaning toward medical societies if there is a difference in opinion between them and the federal government. - If the American Medical Association and the CDC disagreed on the safety of a vaccine, 34 percent of respondents said they would rather accept the American Medical Association’s recommendation, compared to 15 percent who would go with what the CDC recommends. (Seventeen percent said neither, and the rest — 34 percent — weren’t sure.)
- If the American Academy of Pediatrics and the CDC disagree on whether newborns should be given a hepatitis B vaccine, 42 percent were more likely to side with the American Academy of Pediatrics versus 11 percent more likely to follow the CDC’s recommendation. (Sixteen percent said neither, and the remainder — 34 percent — said they weren’t sure.)
→ Check out the latest report from my colleague Lena H. Sun in The Washington Post newsroom that digs even deeper into the poll results. Here’s a chart from Lena’s story that ranks who respondents viewed as most trustworthy providing public health information: | 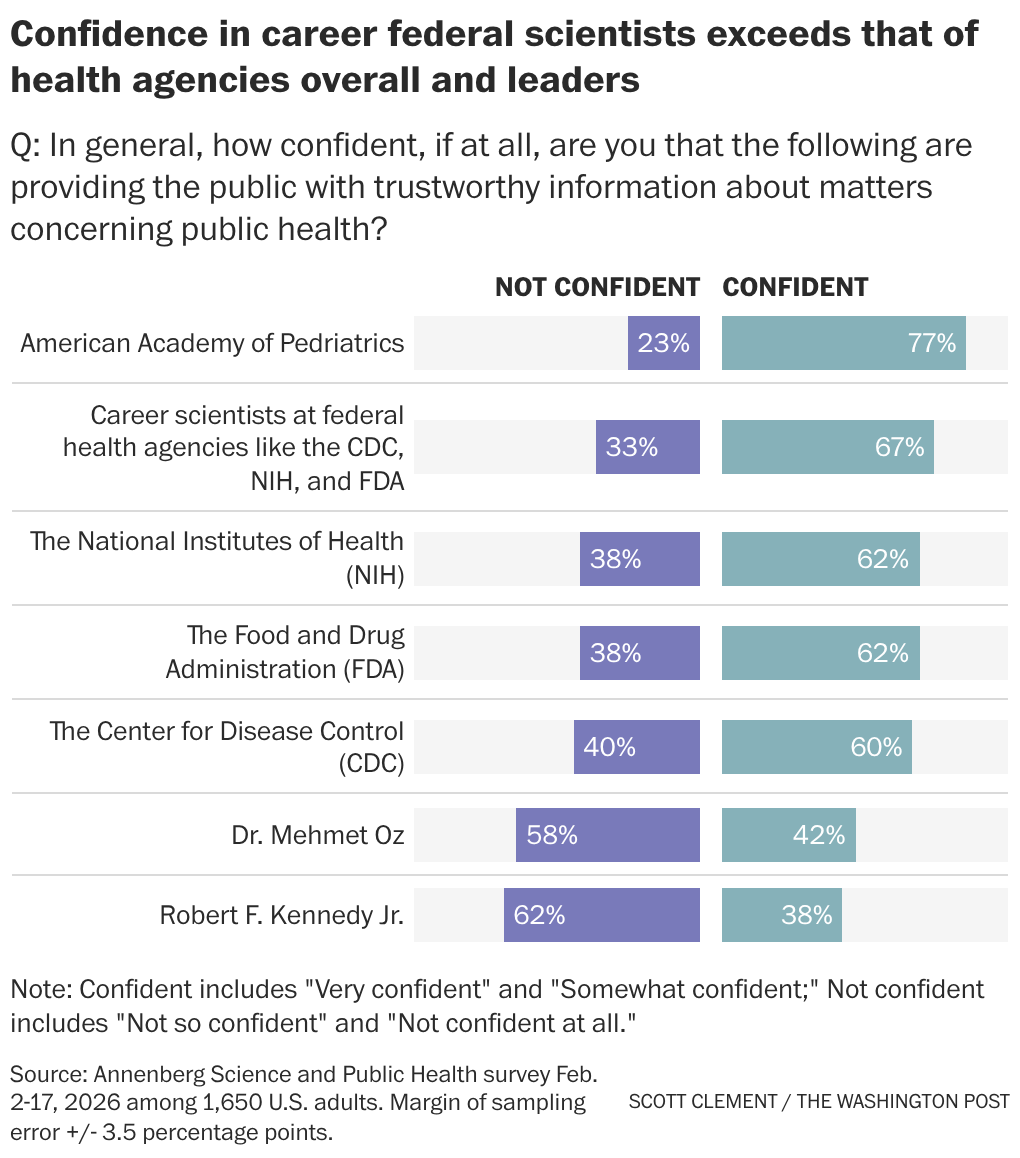 | | Why it matters: The federal government has long established public health recommendations that are ultimately followed by states, public health officials, insurers and medical professionals. However, as the Trump administration overhauls the recommended vaccine schedule, some states and medical societies have broken off and begun offering their own guidance. Top officials, including Health Secretary Robert F. Kennedy Jr., have said the administration is making the changes to regain the public’s trust in the federal government’s public health agencies, but this poll suggests it may be doing the opposite. | | | |  | Industry Rx | The American Hospital Association (AHA) is pressing federal regulators to halt new disclosure requirements that drugmakers are imposing on facilities that participate in the 340B discount drug program. The industry group told the Health Resources and Services Administration, an agency within the Department of Health and Human Services, that the policy would impose “onerous” costs on facilities, and said it violates the federal law governing the discount program by effectively driving up the cost of medicines. Congress created the 340B discount drug program to help hospitals and health clinics that serve a large number of low-income or uninsured patients to stretch limited resources. → Why it matters: The push-and-pull between hospitals and drug companies has been growing more tense as 340B has ballooned into an $81 billion program. The pharmaceutical industry claims that well-off hospitals are able to abuse the program and extract discounts from drugmakers that don’t go toward helping patients. Hospitals, meanwhile, argue that manufacturers are only trying to protect their bottom line. The dispute: In January, the AHA told regulators about a new policy from Eli Lilly requiring facilities that use the 340B program to provide claims-level data for its medications that are dispensed — including by the hospital or clinic, and by contract and in-house pharmacies. “This sweeping expansion of claims data demands will inflict a range of onerous costs and burdens on them,” Chad Golder, AHA’s general counsel, wrote to HRSA. “At best, Lilly’s new requirements will be prohibitively costly for 340B hospitals. At worst, they will be unworkable,” Golder wrote. “Either way, they will prevent hospitals from obtaining the 340B discounts they are owed by statute.” Eli Lilly fired back, and argued that hospitals already provide the data the company is requesting to insurers, thereby rejecting the argument that it’s overly burdensome. “Unfortunately, it seems AHA is more committed to concealing the abuse in the 340B program because hospitals profit from it,” the company said in a statement. Earlier this week, AHA alerted federal regulators to requirements being put in place by Novo Nordisk. Novo Nordisk is echoing Eli Lilly’s arguments to defend the new requirements — set to take effect by April 1 — saying that the information it’s requesting is already compiled by hospitals and provided to third-party organizations. In the letter, the hospital industry group said that HRSA did not reply to its first letter, and said that the agency “cannot remain silent” as additional drug companies implement the transparency rules. “We hope that HRSA will take immediate enforcement action, including the use of civil monetary penalties, against both Lilly and Novo to halt their new policies,” Golder said in a letter dated March 3. “But if HRSA is not going to take such action, it must let 340B hospitals and other stakeholders know that and explain why these new policies are lawful under the 340B statute.” A spokesperson for HHS said the agency is “reviewing these policies and may provide additional comments at a later date.” | | | | |